
You may be familiar with the term “autoimmune,” given increasing attention to chronic inflammation and autoimmune disease in the popular media. Autoimmune means that the body has an immune reaction to substances naturally present in the body. In other words, the immune system attacks “self.” This can happen in most tissues in the body and, accordingly, there are more than 80 autoimmune diseases that have been named and described. Many autoimmune diseases are rare and have been the subject of very limited research. Table 1 lists a small sample of some of the more common autoimmune diseases.
Table 1. Small sample of autoimmune diseases (out of >80)
| Thyroid | Hashimoto Thyroiditis Graves Disease |
| Joints/skin | Psoriasis Vitiligo Rheumatoid Arthritis Ankylosing Spondylitis |
| Central Nervous System | Multiple Sclerosis |
| Pancreas | Type 1 Diabetes |
| Gastrointestinal Tract | Crohn’s Disease Ulcerative Colitis Celiac Disease |
Collectively, though many individual conditions are very rare, autoimmune diseases as a group are actually quite common, affecting perhaps about 7-10% of the population[1], with women being more commonly afflicted. And it seems that the prevalence of autoimmune disease is increasing[2]. Autoimmune diseases can be extremely difficult to diagnose and often present with vague, nonspecific symptoms like fatigue, odd pains, or skin rashes. Given the prevalence, along with the lack of scientific understanding and the vague symptoms, autoimmune diseases have become something of a “catch-all” in the public mind. If an illness goes undiagnosed, is composed of a collection of nonspecific symptoms, people might naturally wonder about “autoimmune.”
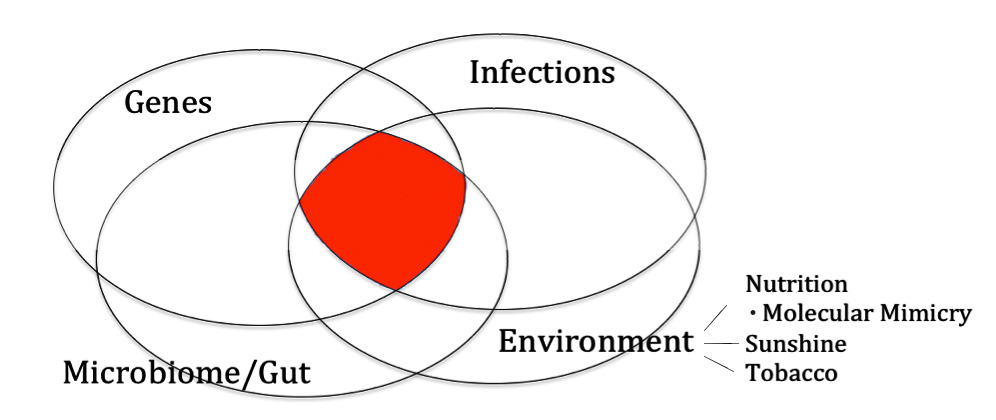
Autoimmune disease is complicated; extraordinarily complicated. In my reading of the literature, there are several intertwined factors that may affect risk of autoimmune disease:
Let’s go through each of these briefly to illustrate why each of these is important.
Many of the patients I have known believe autoimmune disease to be very nearly 100% “in the genes,” particularly those patients with family members who are afflicted. It turns out that genes matter a great deal for some autoimmune diseases but are less important for others.
It seems that the more genetic research has been done for any one disease, the more regions of our genes are found to be involved in risk[3]. HLA genes are particularly prominent in autoimmune disease as they are involved in the body being able to distinguish self from foreign invaders. For example, all celiac patients have one of two HLA genes, though many other genes may be involved[4].
Table 2 shows summary data from identical twin studies. Identical twins, of course, are siblings with exactly the same genes. For those twins where one sibling has the disease, the probability that the other sibling also has the disease is called “concordance.”
Table 2. Autoimmune concordance among identical twins[5]
| Disease | Concordance (Among affected twins, the percent of twins that both get the disease.) |
|---|---|
| Type 1 Diabetes | 13-47% |
| Multiple Sclerosis | 0-50% |
| Celiac Disease | 60-75% |
| Autoimmune Thyroid Disease | 17-22% |
| Psoriasis | 35-64% |
| Crohn’s Disease | 20-50% |
| Ulcerative Colitis | 6-18% |
| Lupus | 11-40% |
| Rheumatoid Arthritis | 0-21% |
| Ankylosing Spondylitis | 50-75% |
Some conditions, like celiac disease and ankylosing spondylitis, seem to have very strong genetic components. But the majority of autoimmune diseases only appear in both siblings in a minority of identical twins. The risk of getting autoimmune conditions like multiple sclerosis, lupus, rheumatoid arthritis, ulcerative colitis, and type 1 diabetes clearly must be strongly affected by something other than genes.
In other words, for many of these autoimmune conditions, having high-risk genes may be a necessary but not sufficient reason for a person to get the disease. It’s something more than just genes.
It should come as no surprise that certain infections are linked to some autoimmune diseases. What does your body do in response to infections? It revs up the immune system to fight the invaders. In some cases infections may actually lower the risk of autoimmune disease, and in some cases they may cause autoimmune disease[6].
Infections can be damaging, though, too. Several autoimmune diseases have been linked to specific infections. Sometimes, it seems that chronic infection and inflammation from a viral or bacterial invader can actually precipitate immune system confusion at some point, triggering the start of the autoimmune process[6]. People who have been exposed to Epstein Barr Virus (EBV), which causes mono, have a higher rate of multiple sclerosis[7]. Those who have been infected with EBV in adolescence or later in life have a 30 times higher risk of multiple sclerosis compared to those who have never had Epstein Barr Virus[7]. That’s a big number.
How does it work? We know that there are parts of these infections that look a lot like parts of our body. When the immune system attacks the infection, it can cross-react and attack the similar parts of our own body, not being able to distinguish between the invader and “self.” This is called molecular mimicry. The molecules mimic each other, in a sense. For example, this can happen when the bacteria that causes strep throat (Streptococcus pyogenes), leads to immune system cross-reaction with heart muscle, causing rheumatic fever. This is a main reason why we give antibiotics for strep throat. Similarly, Ankylosing Spondylitis has been linked to Klebsiella pneumoniae, and Guillain-Barré has been linked to Campylobacter jejuni. There are dozens of links between various autoimmune conditions and viruses or bacteria[9].
If there’s an important genetic component and infections might play an important role, are autoimmune diseases basically just “luck of the draw” illnesses? No. That’s clearly not the whole picture, based on data related other environmental exposures.
Multiple sclerosis (MS) rates vary widely throughout the world. When I tell you that the farther away a population is from the equator, the higher the rate of multiple sclerosis[10], how do you make sense of that? How about the fact that if you were born in November you have a slightly lower rate of MS and if you were born in May you have a slightly higher risk of getting MS than what otherwise would be expected by random chance[11]? How are these connected? Does cold weather somehow cause MS, or some other seasonal factor?
And we now know that if migrants from a high risk area move to a low risk area, their risk becomes lower when compared to that of their homeland population[12].
We obviously have to consider more than genes and viruses. There’s some other environmental exposure that matters. By environment, I mean exposures related to food, exercise, tobacco, weather, environmental chemicals, X-rays, or basically anything to which we’re exposed. In the mystery of why multiple sclerosis is more common the farther from the equator you go, we can find an answer in vitamin D. Lower vitamin D levels are linked to higher the risk of MS[13], and a higher the risk of exacerbations if you have the illness[14]. People far from the equator have lower vitamin D levels. And babies born in the northern hemisphere in November compared to May have been in utero when their mom is more likely to have had better vitamin D status.
We make vitamin D when our skin is exposed to sunlight, so you can see how being far away from the equator might be reasonably linked to disease risk. We also know now that vitamin D is involved in immune system function[15]. It’s important to note that taking vitamin D supplements have not been shown to be helpful for multiple sclerosis[16]. You can’t take a nutrient supplement and get the same benefit for chronic disease, which is a principle that has been proven to be true many times in many studies.
What about nutrition? It turns out that there’s a striking correlation between a population’s milk consumption and how much multiple sclerosis is in the population[17]. The more cow’s milk, the more multiple sclerosis.
Molecular mimicry may be playing a role. Multiple sclerosis is a disease of damage to the nerve sheaths, the important covering to our nerve cells. It turns out that there’s a protein in cow’s milk that looks a lot like a protein on our nerve sheaths[18]. Our immune system reacts to both the cow’s milk protein and the nerve sheaths.
The link between dairy intake and autoimmune disease and molecular mimicry doesn’t stop with multiple sclerosis. Type 1 diabetes, a disease where the immune system attacks the pancreas, has also been linked to dairy. There is a striking correlation between high dairy consumption in a population and high rates of type 1 diabetes[19]. Patients with the disease also have a strong immune response to proteins in milk that “mimic” components of the pancreas[20] [21]. Type 1 diabetes is, by definition, a disease resulting from destruction of the insulin producing cells of the pancreas. Why does the immune system attack both the proteins in milk that “mimic” pancreas components and the pancreas itself? Are exposures to milk proteins “triggering” the autoimmune process? This has been an ongoing hypothesis.
It may be that the timing of exposure to milk also plays an important role. The timing of when an infant starts consuming solid foods seems to affect risk[22]. Breast milk appears to be protective[23]. One hypothesis has been that if infants are exposed to cow’s milk proteins too early, the proteins get through the wall of the gut and then play a role in initiating the autoimmune process.
It turns out that animal studies support this idea. There is a type of rat that has been bred to have a high rate of type 1 diabetes and, at the time of weaning, exposure to cow’s milk protein leads to a dramatically increased incidence of type 1 diabetes[24]. Take away the milk protein and you can take away most of the diabetes.
But it gets even more complicated because it’s not just related to cow’s milk. It turns out you can also trigger type 1 diabetes in this animal model with gluten, the protein found in wheat, barley, and rye. If researchers withhold gluten at the time of weaning in mice bred to get diabetes, they can quite strikingly prevent diabetes[25].
Gluten, of course, is also strongly linked to celiac disease. In fact, celiac disease does not exist without gluten. When exposed to gluten, the immune system goes a bit haywire in the intestinal wall and the resulting inflammation ends up greatly harming the intricate surface of the intestine. The disease is cured by removing gluten from the diet. In related conditions, we also know that gluten can cause something called gluten ataxia, a very serious debilitating disease causing loss of coordination and muscle function, and dermatitis herpetiformis, a skin rash. Both of these, based on our current understanding, are rare.
Of course, it’s not just cow’s milk and gluten that have been implicated in autoimmune disease. We know that risk of inflammatory bowel disorders, crohn’s colitis and ulcerative colitis, has been linked to infant feeding practices (breastfeeding being protective), cow’s milk, low intake of fiber and vegetables, and increased meat and added fats in the diet[26] [27]. Thyroid conditions are linked to iodine intake[28]. Not enough iodine and too much iodine[29] both can be problems.
Partly from the research on celiac disease, we now know that part of the problem is a breakdown in the barrier in the intestine. It involves what is commonly known as “leaky gut.” The ‘seal’ between the cells that line the gut, which keeps the outside food outside, break apart. Once the barrier is disrupted, incompletely digested material gets behind the first layer of cells in the gut. This can cause problems.
And if autoimmune diseases sometimes cluster together is it possible that “leaky gut” is playing a role common to many of these illnesses? Type 1 diabetes, celiac disease, and thyroid disease can cluster together in the same individuals, for example[30].
Of course, the health of the intestine is related to the health of the bacteria that live in the intestine. And despite the rampant claims and products and pills and potions found on the internet for the buzzwords “leaky gut,” “inflammation” and “microbiome,” our current understanding of which bacteria exactly are good, what exactly they do, and how to best support them is in the early stages of research. Generally, we know that fiber and unrefined plant foods like leafy greens help to maintain a healthy diversity of beneficial bacteria in your intestine. This is consistent with research showing that Western diets high in meat, dairy, saturated fats, and low in fiber, fruits and vegetables are linked to some autoimmune diseases and increases inflammation in general.
Figure 1. The Autoimmune Formula
So what do you take away from all this? I wish I could offer a simple explanation that answers the question about what causes autoimmune disease. But I can’t. I don’t think the science supports making blanket statements about autoimmune disease, but there are some key lessons we’ve seen so far that may be helpful.
As far as controllable factors, it seems that cow’s milk is, by far, the food most commonly linked to autoimmune disease. I suggest to my patients that they avoid cow’s milk products in all their forms. Second, avoid other components of a “western”-style diet, which means avoiding meat, added oils, and dairy (already mentioned) and including plenty of fiber and green, yellow and orange vegetables. Third, infant nutrition and breastfeeding may play a role, so I encourage moms to do everything they can to breastfeed. Fourth, get outside and get sun (without burning) and stay active regularly. Fifth, don’t smoke (I didn’t discuss this earlier, but smoking has been linked to some autoimmune disease). Sixth, include some sea vegetables in the diet now and then or use iodized salt to ensure iodine intake.
You may have noticed I didn’t mention gluten in my recommendations. I think it reasonable to avoid gluten, particularly if you have autoimmune disease or are at risk, but you may want to discuss with your doctor being tested for celiac before embarking on a gluten-free life. Consider getting tested for celiac disease if you have autoimmune disease or think you generally have reactions to gluten. Despite the attention heaped on gluten, it is not as strongly implicated in a broad array of autoimmune disease as is cow’s milk. It is difficult to avoid gluten 100% of the time and for most people, I’m not quite convinced it’s necessary. I have two full chapters on gluten in The China Study Solution (which is The Campbell Plan in hardcover).
By adhering to these strategies will everyone avoid all autoimmune disease? Of course not. I believe it to be clear that there is more to autoimmune disease than nutrition. But I also believe that nutrition is likely to be the single greatest modifiable risk factor that affects autoimmune disease. And this isn’t just about prevention. There is now a smattering of evidence that diet can be an effective part of treating autoimmune disease.
In various publications, diet and lifestyle treatments have shown benefit in the treatment of rheumatoid arthritis, psoriasis, Crohn’s colitis, multiple sclerosis, and even type 1 diabetes. Once people are diagnosed with ulcerative colitis, we now know that consuming a more Western diet has been linked to worse outcomes. And we know, of course, that avoiding gluten is the definitive treatment for celiac disease. Importantly, diet and lifestyle can also improve risks of related chronic diseases (many autoimmune conditions, for example, are linked to a higher risk of heart disease). So what have we got to lose in considering nutrition as part of the autoimmune “conversation”? How about some cholesterol? Or some weight? And maybe we’ll find powerful success for some of us with autoimmune disease along the way.
Copyright 2024 Center for Nutrition Studies. All rights reserved.